A securities class action has been filed Inovio Pharmaceuticals, Inc. (INO) on behalf of a class consisting of all persons and entities other than Defendants that purchased or otherwise acquired Inovio securities between October 10, 2023 through December 26, 2025. This case has been filed in the USDC – SDNY.
Inovio is a biotechnology company focused on the discovery, development, and commercialization of DNA medicines to treat and protect people from diseases associated with, inter alia, human papilloma virus (“HPV”). The Company’s DNA medicines are comprised of two components: (i) DNA plasmids, which are small circular DNA molecules that purportedly work like software that the body’s cells can download to produce specific proteins to target and fight disease; and (ii) its proprietary investigational medical device, “CELLECTRA,” which it uses to help its DNA medicines enter the body’s cells for purported optimal effect.
Inovio’s lead product candidate is INO-3107 for the treatment of recurrent respiratory papillomatosis (“RRP”), a life-long, rare disease of the respiratory tract caused by HPV infection. At all relevant times, Defendants touted the prospects of the U.S. Food and Drug Administration (“FDA”) granting accelerated approval and/or priority review for the Biologics License Application (“BLA”) of INO-3107 for the treatment of RRP (the “INO-3107 BLA”). Defendants also touted their ability to complete rolling submission of the INO-3107 BLA by the second half of 2024. In so doing, Defendants consistently and repeatedly indicated to investors that Inovio was rapidly approaching its transition into a commercial-stage company-one with a lead product asset that, once approved, would fill an unmet medical need and significantly improve the safety or effectiveness of current RRP treatments. The commercial implications of this prospect, which Defendants consistently highlighted throughout the Class Period, were of the upmost importance to investors and analysts, and formed a core part of the Company’s overall investment thesis.
Simultaneously, while disseminating these positive statements to the market, throughout the Class Period Defendants conducted numerous offerings of Inovio’s securities, reaping profits of tens of millions of dollars per offering.
Throughout the Class Period, Defendants made materially false and misleading statements regarding the Company’s business, operations, and prospects. Specifically, Defendants made false and/or misleading statements and/or failed to disclose that: (i) manufacturing for Inovio’s CELLECTRA device was deficient; (ii) accordingly, Inovio was unlikely to submit the INO-3107 BLA to the FDA by the second half of 2024; (iii) Inovio had insufficient information to justify the INO-3107 BLA’s eligibility for FDA accelerated approval or priority review; (iv) accordingly, INO-3107’s overall regulatory and commercial prospects were overstated; and (v) as a result, Defendants’ public statements were materially false and misleading at all relevant times.
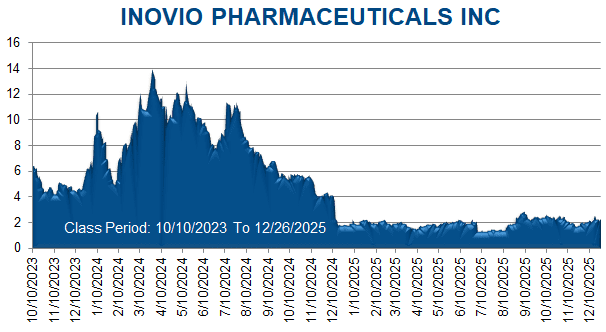
The truth began to emerge on August 8, 2024, when, during post-market hours, Inovio issued a press release reporting its financial results and recent business highlights for the second quarter of 2024. Therein, Defendants revealed that Inovio expected to submit the INO-3107 BLA to the FDA in mid-2025-representing an approximate full-year delay from Defendants’ initially projected mid-2024 submission timeline-because of “a manufacturing issue” with a component of the CELLECTRA device.
On this news, Inovio’s stock price fell $0.27 per share, or3.1%, to close at $8.44 per share on August 9, 2024.
Then, on December 29, 2025, during pre-market hours, Inovio issued a press release announcing that the FDA had accepted the INO-3107 BLA on a standard rather than accelerated review timeline. Defendants advised that the FDA had indicated that the Company did not submit adequate information to justify eligibility for accelerated approval. Defendants further advised that Inovio does not plan to seek approval under the standard review timeline and would request a meeting with the FDA to discuss how it may still pursue accelerated approval.
On this news, Inovio’s stock price fell $0.56 per share, or 24.45%, to close at $1.73 per share on December 29, 2025.