A securities class action has been filed against ImmunityBio, Inc. (IBRX) on behalf of persons or entities who purchased or otherwise acquired publicly traded ImmunityBio securities between January 19, 2026 through March 24, 2026. This case has been filed in the USDC – CDCA.
On January 23, 2026, ImmunityBio issued a press release “announc[ing] updated Phase 2 clinical results from QUILT 3.078 (NCT06061809), evaluating a chemotherapy-free combination immunotherapy regimen in patients with second-line recurrent or progressive glioblastoma (GBM), as well as patients treated under single-patient INDs (spINDs) across first- to third-line disease.” Among other results, the press release disclosed that “median overall survival”—the primary endpoint for the trial—”has not yet been reached[.]”
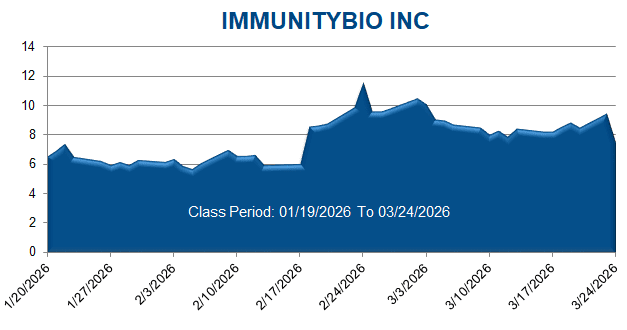
On this news, ImmunityBio’s stock price fell $0.89 per share, or 12.13%, to close at $6.45 per share on January 23, 2026.
Then, on March 24, 2026, a warning letter to ImmunityBio from the U.S. Food and Drug Administration (“FDA”), dated March 13, 2026, became public. In the letter, the FDA asserted that, despite previous warnings from the FDA about the accuracy of the Company’s promotions, “ImmunityBio continues to promote Anktiva”—the Company’s bladder-cancer drug—”in a similar misleading manner”, including in television advertisements and on a January episode of Sean Spicer’s podcast.
On this news, ImmunityBio’s stock price fell $1.99 per share, or 21.12%, to close at $7.41 per share on March 24, 2026.