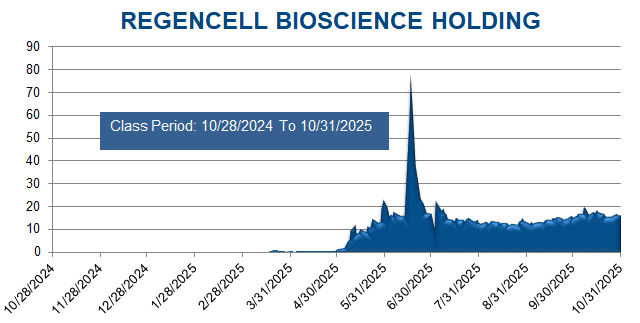
A securities class action has been filed against Regencell Bioscience Holdings Limited (RGC) on behalf of a class consisting of all persons and entities other than Defendants that purchased or otherwise acquired Regencell securities between October 28, 2024 through October 31, 2025. This case has been filed in the USDC – MD.
Regencell is a purported early-stage bioscience company focused on the research, development, and commercialization of traditional Chinese medicine (“TCM”) for the treatment of attention-deficit/hyperactivity disorder (“ADHD”) and autism spectrum disorder (“ASD”)— two disorders generally considered incurable under current medical consensus. The Company asserts that while “[c]urrently available drugs and treatments in the markets for ADHD and ASD aim to suppress or alleviate symptoms,” its “TCM formula aim[s] to treat the fundamental cause of neurocognitive disorders.”
The complaint alleges that throughout the Class Period, Defendants made materially false and misleading statements regarding the Company’s business, operations, and compliance policies. Specifically, Defendants made false and/or misleading statements and/or failed to disclose that: (i) Regencell was vulnerable and/or subject to market manipulation; (ii) the resulting volatility in the market for the Company’s ordinary shares exposed Regencell’s investors to significant financial risk; (iii) all the foregoing subjected Regencell to a heightened risk of regulatory and/or governmental scrutiny and enforcement action, as well as significant legal, monetary, and reputational harm; and (iv) as a result, Defendants’ public statements were materially false and misleading at all relevant times.